
Pembrolizumab is currently approved by the FDA for use in combination with chemotherapy for the treatment of patients with locally recurrent unresectable or metastatic TNBC whose tumors express PD-L1 (CPS ≥10) as determined by an FDA-approved test. “These results provide further support for pembrolizumab in combination with chemotherapy as the new standard-of-care treatment regimen for patients with locally recurrent, unresectable or metastatic TNBC whose tumors express a PD-L1 CPS of 10 or more,” lead study author Javier Cortés, MD, PhD, the head of breast cancer and gynecological cancers at Hospital Universitario Ramón y Cajal, in Madrid, Spain, said in a presentation of the data. The 12-month PFS rate was 39.1% with pembrolizumab and 23.0% with placebo/chemotherapy. Moreover, the median progression-free survival (PFS) elicited by the combination was 9.7 months vs 5.6 months in patients with a CPS of at least 10 (HR, 0.66 95% CI, 0.50-0.88).

The 18-month OS rates were 58.3% with pembrolizumab vs 44.7% with placebo/chemotherapy. Results from the study, which were presented at the 2021 San Antonio Breast Cancer Symposium, showed that the combination of pembrolizumab and chemotherapy yielded a median overall survival (OS) of 23.0 months vs 16.1 months with chemotherapy alone in patients with a CPS of 10 or higher (HR, 0.73 95% CI, 0.55-0.95 P =.
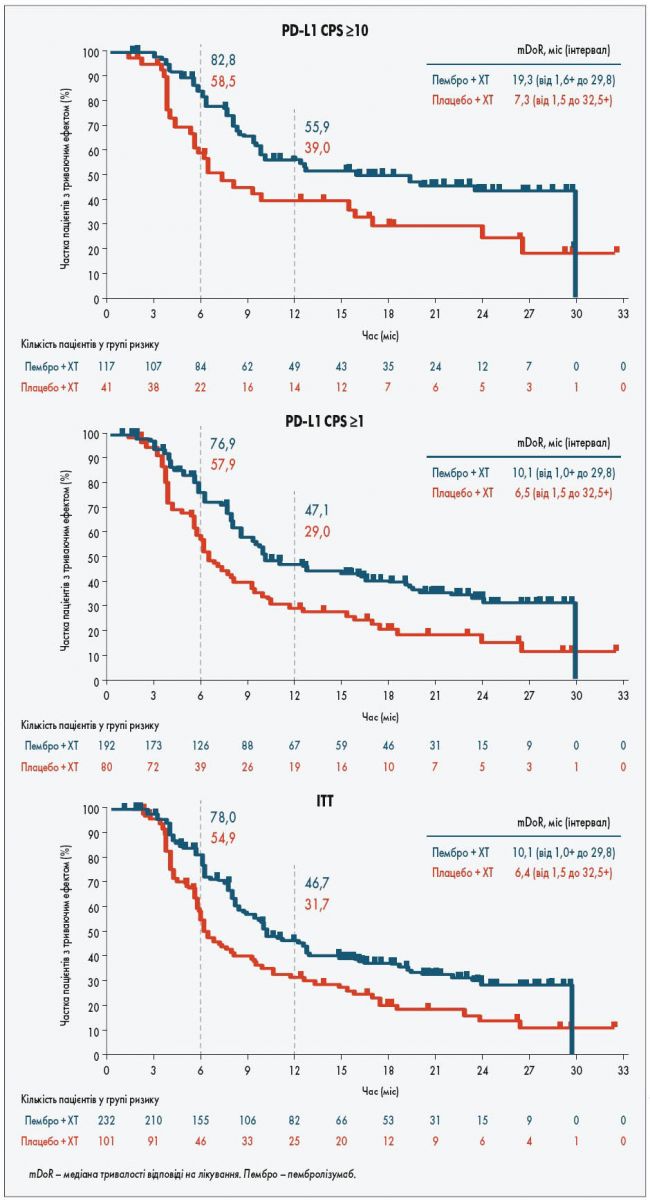
1įurthermore, a PD-L1 CPS of at least 10 was found to be a reasonable cut-off to define the population of patients expected to derive the most benefit from this regimen.
#KEYNOTE 355 TRIAL#
Treatment with pembrolizumab (Keytruda) and chemotherapy yielded a statistically significant and clinically meaningful survival benefit compared with chemotherapy alone in a population of patients with previously untreated, locally recurrent, inoperable, or metastatic triple-negative breast cancer (TNBC) with a PD-L1 combined positive score (CPS) of 10 or more, according to updated findings from the phase 3 KEYNOTE-355 trial (NCT02819518).
